My approach: structured research while staying curious

I first came into contact with the power of this approach during my PhD research for drinking water company Oasen into the inhibition of nitrification by iron removal.
At the start of this research, there was an almost unanimous view in the scientific community and drinking water sector that iron removal under oxygen-saturated conditions and at a slightly alkaline pH - the conditions in Oasen's purification plants - takes place entirely chemically.
Of course, that was also an unwavering starting point for me at that time.
The central research question concerned the effect on nitrification of pre-oxidation of iron before filtration, i.e. whether iron ends up on the filter as (chemically formed) iron flakes or in dissolved, reduced form. A fully automatic test installation with 20 columns of my own design enabled me to simultaneously investigate six hypotheses for several months in parallel and of course in duplicate. Before the actual research at the groundwater purification of Oasen Lekkerkerk, I had tested the installation extensively and fully completed and evaluated all research protocols in a test hall of the TU Delft. Early 2009, the research at the groundwater location Lekkerkerk of Oasen started.
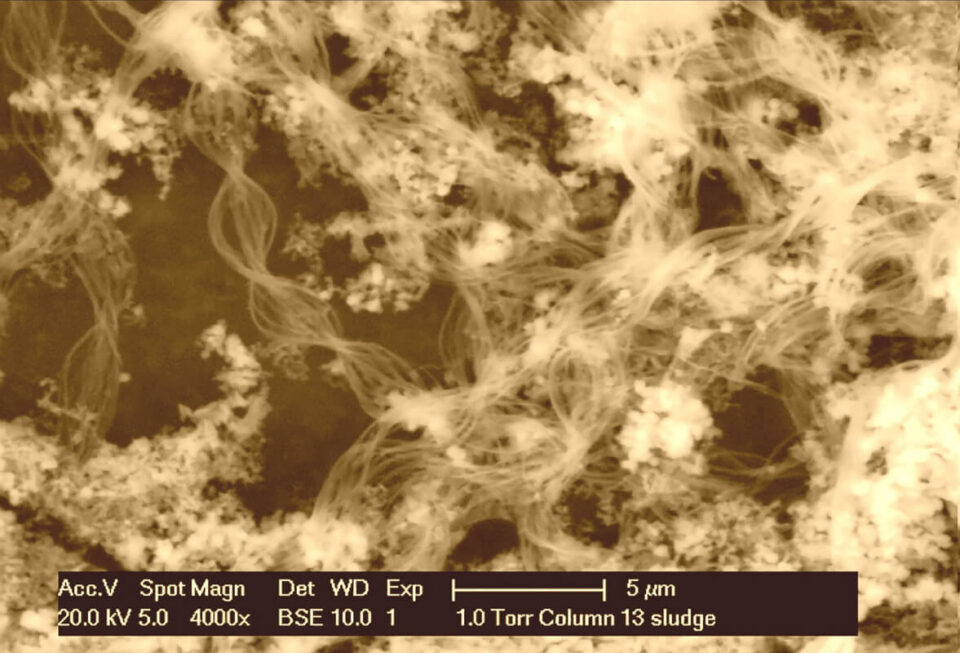
The results of the research, which had gone entirely according to plan, however, seemed to be completely off the mark when evaluated. Where, as expected, neat, chemically formed iron flakes were formed during pre-oxidation of iron in the water phase at TU Delft, tangles of rust-coloured threads were formed at Oasen Lekkerkerk. After a few weeks of nagging doubt and thorough checking of the experimental conditions, the conclusion could only be that the experiments had gone correctly and that I had observed something exceptional at the Oasen Lekkerkerk groundwater purification plant. Parallel molecular research, light and electron microscopic recordings and additional calculations provided further confirmation of biological iron oxidation far beyond the usual range.
I decided to repeat the experiments at the groundwater location Oasen Lekkerkerk at oxygen saturation and to investigate a new variable, the pH (acidity). The iron oxidation in groundwater filters appeared to be mainly biological (by Gallionella spp.) over a much larger range in oxygen and pH than previously published (up to pH 7.7 under full oxygen saturation). These results were confirmed by molecular measurements in full-scale production plants: the practical filters were also teeming - under oxygen-saturated conditions and pH well above 7 - with iron-oxidizing Gallionella-bacteria (click here for the peer-reviewed article in the renowned scientific journal Water Research.
How could the results between TU Delft and Oasen Lekkerkerk differ so much? Here again it became clear that a devil was hidden in the details. An important, if not crucial difference was the temperature during the experiments, 20-25 °C in the test hall of the TU Delft, 12 °C at the groundwater purification of Oasen Lekkerkerk. This seemingly small difference theoretically appeared to explain a delay of the chemical iron oxidation by a factor of 7. Seemingly small differences between laboratory and practical research appeared to give decisively different results. The most important lesson for me as an applied scientist was to always let systematic and careful observation prevail over conventions. A theoretical insight is only correct if it has withstood the test of practice.
Images 2007 TU Delft


Images 2008-2009 Oasen Lekkerkerk